PRACTICE FORUM
Reuse of disposable gowns for COVID-19 personal protective equipment purposes in times of scarcity: results of an experimental study with hydrogen peroxide vaporization
Roy J. Pelzer*, Elke JP Magdeleyns, Paul HM Savelkoul, Lieke B van Alphen and Wil C van der Zwet
Department of Medical Microbiology, Maastricht University Medical Center, and Care and Public Health Research Institute (CAPHRI), Maastricht University, Maastricht, The Netherlands
Abstract
During the coronavirus disease 2019 (COVID-19) pandemic, many health organizations faced shortages of personal protective equipment for their personnel. In case of extreme urgency, re-using disposable materials might offer a temporary solution. Hydrogen peroxide vaporization (HPV) has been used for disinfection of patient rooms for more than a decade. We investigated HPV as a method for disinfecting disposable gowns. After HPV, gowns proved to be free of bacteria and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA while their functionality and water-repellency remained intact. We conclude that, in case of emergency and lack of available alternatives, HPV is a suitable and relatively inexpensive method for one-time reuse of disposable gowns.
Keywords: Hydrogen peroxide (H2O2), disinfection, personal protective equipment, COVID-19
Citation: Int J Infect Control 2021, 17: 21090 – http://dx.doi.org/10.3396/ijic.v17.21090
Copyright: © 2021 Roy J. Pelzer et al. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material for any purpose, even commercially, provided the original work is properly cited and states its license.
Received: Received: 07 December 2020; Accepted: 15 January 2021; Published: 16 March 2021
Competing interests and funding: The authors declare no conflicts of interest. This research received no external funding.
*Roy J. Pelzer, Department of Medical Microbiology, Maastricht University Medical Center, P. Debyelaan 25, 6229 HX Maastricht, The Netherlands, Email: roy.pelzer@mumc.nl
The World Health Organization recommends using a medical mask, eye protection, gloves and a clean non-sterile long-sleeved gown as personal protective equipment (PPE) in coronavirus disease 2019 (COVID-19) patient management (1). The COVID-19 global pandemic has resulted in an overwhelming need of adequate PPE for healthcare workers worldwide, as most of them are single-use and disposable (2, 3). In order to address these shortages, many inventive initiatives have been undertaken to produce PPE locally or nationally, such as handmade 3D-printing (4). Furthermore, there is a growing amount of publications on decontamination and reuse of disposable PPE, mainly focusing on protective masks, but there are concerns regarding safety aspects (5). However, the Centers for Disease Control and Prevention (CDC) recently released information about medical mask decontamination and reuse as a ‘crisis capacity strategy to ensure continued availability’, and hydrogen peroxide vaporization (HPV) was listed as one of the most promising potential methods (6).
Hydrogen peroxide vaporization has been effectively used for disinfecting contaminated environmental surfaces in the hospital (7). It is also effective against syndrome coronavirus 2 (SARS-CoV-2) and has been used successfully in sterilizing N95 respirator masks (8, 9).
Background
In this experimental study, we investigated the effect of HPV on disposable gowns, used in care for COVID-19 patients, on functionality, water repellency and microbiological safety. This method proved to be very effective and may offer a solution for shortages of PPE in hospitals situated in low and middle-income countries in general.
Methods
Materials
Five disposable isolation gowns from two suppliers that had been used in COVID-patient care were investigated for bacterial decontamination experiments: Q-plus (EMD 95144, n = 2) and Emdamed (EMD 95033, n = 3) (both from Emdamed, Berkel en Rodenrijs, Netherlands). For viral decontamination experiments, a new gown was used (Emdamed). Gowns were hung on a rack and placed in a room of 39.7 m3 in the hospital building. This experimental room was not used for patient care, but the ventilation rate was equal to patient rooms.
Methods
Hydrogen peroxide vaporization
The rack with gowns was placed in the centre of the concerning room (total content 27.6 m3 underneath the ceiling or 39.7 m3 if the space above the ceiling is added to the volume). The distance between the gowns was approximately 15 cm, and their bottoms were approximately 50 cm above ground level. The H2O2 detector was placed in a corner of the room. Subsequently, the room was HPV treated with 400 mL Nocolyse, containing 60 mL H2O2 and 17 ppm silver ions per litre, for 25 min at a mean fixed rate of 16 mL/min, followed by a dwelling phase of approximately 2 h. All was performed according to the instructions of the manufacturer (Oxy`pharm, Champigny-sur-Marne, France).
Microbiological testing
Testing bactericidal effectiveness. For this experiment, five gowns were used that had been used in daily patient care; they were not deliberately seeded with bacteria before testing. Prior to HPV, from each gown a 1 × 1 cm piece was cut out in a safety-cabinet, using sterile forceps and sterile scissors. After HPV, three random pieces from the same size from each gown were obtained in the same way; also some pieces of sleeves and fixation-bands were sampled. All cut-out pieces were placed in sterile tubes containing fluid thioglycollate medium without indicators, using sterile forceps. Tubes were incubated at 35°C for 72 h and subsequently assessed for turbidity, indicating microbial growth.
Testing SARS-CoV-2 reduction. For COVID-19 diagnostics in our hospital, material from plain sterile swabs with twisted applicator (COPAN Diagnostics Inc, Murietta, CA, USA) from combined nasopharynx-throat is transferred to universal viral transport medium (VTM, Becton Dickinson, Franklin Lakes, NJ, USA) and subsequently tested with polymerase chain reaction (PCR). The remaining material is stored at −80°C. For our experiment, the original material from a strong positive sample [cycle threshold (CT) 20, matching a viral load of 108 RNA copies/mL], which had been stored at 4°C for 2 days, was used. On a new gown directly taken from the packaging, four circles with an approximate diameter of 2 cm were marked on the chest part. From the positive sample, 400 μL were added to the centre of each marked circle (Fig. 1). After drying on air for 10 min, each circle was swabbed with a dry sterile swab, which was subsequently washed out in 2 mL VTM. After this, the gown was hung on the rack in the experimental room and HPV treatment was performed. Post-treatment, the specified locations were sampled again in the same manner to recover any SARS-CoV-2 RNA on the gown material.
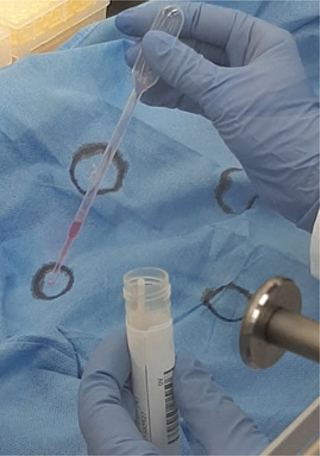
Fig. 1. Eluate from SARS-CoV-2 specimen is spiked on gown.
Laboratory detection of SARS-CoV-2 was performed with a clinically validated real-time polymerase chain reaction (RT-PCR) assay. Technical validation was performed using dilution ranges of a sample with a known viral load obtained from the Dutch Public Health Agency (RIVM). First, RNA was extracted from 90 μL fluid with the MagNA Pure 96 system (Roche, Germany) and eluted in 100 μL of elution buffer from the small volume DNA/RNA extraction kit (Roche). Ten microlitres of the eluted sample was added to a 25 μL RT-PCR plate. Targets RdrP-gene and E-gene were used, which had been validated previously for routine diagnostic use (10). Thermal cycling was performed on a Quantstudio 5 (Applied Biosystems, Waltham, MA, USA). Oligonucleotides were synthesized and provided by Tib-Molbiol (Berlin, Germany) or by Biolegio (Nijmegen, Netherlands).
Functional testing after hydrogen peroxide vaporization
Permeability. A part of the gown was placed horizontally by hand and 10 mL water was poured on it. This was followed by a 3-min observation period to assess whether the inside of the gown remained dry.
Functionality. Two independent observers compared the HPV-treated gowns manually and visually with new gowns, regarding elasticity, structure, firmness and functionality.
Results
Hydrogen peroxide vaporization
The Nocolyse dose was 10.07 mL/m3 and resulted in a peak concentration of 115.8 ppm H2O2 after 25 min. The ventilation rate of the room was 2 air changes per hour.
Microbiological testing
Bactericidal effectiveness. All tubes with material obtained before HPV were turbid, indicating microbial growth. All tubes with material obtained after HPV were clear.
SARS-CoV-2 RNA reduction. PCR results from swabs from the four circles obtained before HPV were positive. Cycle threshold values were 29.5, 28.5, 29.4 and 29.8, respectively. PCR results after HPV were all negative, which showed evidence of effective HPV treatment.
Functional testing
Permeability. The gown material remained water resistant at room standard temperature and pressure for 3 min. The interior of the gown remained dry.
Functionality. After HPV, no visual or tangible abnormalities, including elasticity, structure and firmness, were observed.
Discussion
According to the manufacturer, a minimum of 40 ppm H2O2 is necessary. In our experiment, we exceeded this minimum almost three times, and this resulted in undetectable SARS-CoV-2 after HPV. However, only five gowns were tested and the maximum number of gowns that can be decontaminated simultaneously in a defined room was not further investigated.
Our decontamination experiment turned out to be successful since no remainders of SARS-CoV-2 were detectable after HPV. The higher CT values observed for the samples after swabbing compared to the original material used to spike can be explained by two distinct reasons: (1) the amount of spiked material taken up by the swab, as mentioned before and (2) the dilution factors of the swabbing experiment compared to the original experiment: 400 μL the original material was used and allowed to dry on the gown. After extensive swabbing of the spiked area, the swab was placed in 2 mL VTM (five-fold dilution). Of this material, 90 μL was used as input for RNA extraction (~20-fold dilution) and eluted in 100 μL elution buffer of which 10 μL was used for PCR (10-fold dilution). This gives a total 1,000-fold dilution, which would correspond with a 9–10-fold change in CT value. The samples without HPV treatment showed a CT value 9–10 fold higher than the original material, which indicated that the majority of the spiked material was recovered by the extensive swabbing.
It can be argued whether swabbing was the correct sampling strategy. We considered the possibility that viral material remained clinched to the material. We did not repeat the experiment because our conclusion was that if intensive swabbing had not been able to remove viral material, it is certainly not a risk for nosocomial transmission when the HPV-treated gown would be reused.
In this study, only small numbers of gowns were tested, but the experiments were performed in the peak period of the COVID-19 crisis in the Netherlands under the threat of acute shortages of PPE for our healthcare workers. Nevertheless, the results may be of value for colleagues in the same situation. We concluded that hydrogen peroxide vaporization is a safe method for re-using gowns and may contribute to combat these shortages.
References
- World Health Organization. Infection prevention and control during health care when COVID-19 is suspected: interim guidance. Available from: https://www.who.int/publications/i/item/10665-331495.
- Ranney ML, Griffeth V, Jha, AK. Critical supply shortages – the need for ventilators and personal protective equipment during the Covid-19 pandemic. New Engl J Med 2020; e41: 1–3. doi: 10.1056/NEJMp2006141
- Rowan NJ, Laffey JG. Challenges and solutions for addressing critical shortage of supply chain for personal and protective equipment (PPE) arising from Coronavirus disease (COVID19) pandemic – case study from the Republic of Ireland. Sci Total Environ 2020; 725: 138532. doi: 10.10161/j.scitotenv.2020.138532
- Ishack S, Lipner SR. Applications of 3D printing technology to address COVID-19 related supply shortages. Am J Med 2020; 369: m1577. doi: 10.1136/bmj.m1577
- Rimmer A. Covid-19: experts question guidance to reuse PPE. BMJ 2020; 369: m1577. doi: 10.1136/bmj.m1577
- Centers for Disease Control and Prevention. Decontamination and reuse of filtering facepiece respirators. Available from: www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/decontamination-reuse-respirators.html.
- Weber DJ, Rutala W, Anderson DJ, Chen LF, Sickbert-Bennett EE, Boyce JM. Effectiveness of ultraviolet devices and hydrogen peroxide systems for terminal room decontamination: focus on clinical trials. Am J Infect Control 2016; 44: e77–84.
- Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of coronavirus on inanimate surfaces and their inactivation with biocidal agents. J Hosp Infect 2020; 104: 246–51.
- Perkins DJ, Villescas S, Wu TH, Muller T, Bradfute S, Hurwitz I, et al. COVID-19 global pandemic planning: decontamination and reuse processes for N95 respirators. Exp Biol Med 2020; 245(11): 933–939. doi: 10.1177/1535370220925768
- Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu DK, et al. Detection of 2019 novel coronavirus (2019-nCOV) by real-time RT-PCR. Euro Surveill 2020; 25: 2000045. doi: 10.2807/1560-7917.ES.2020.25.3.2000045